Penyertaan Staf PKTAAB Meriahkan Sambutan Hari Pekerja 2026 Bersama Kontinjen USM
May 01, 2026
September 30 2025
In the screening of candidate anticancer drugs, the preclinical phase is a crucial step in bridging the translational gap to test the efficacy and toxicity of the candidate drugs. Despite the rapid and extensive advancements in researching and discovering potential anti-cancer agents, only one out of ten candidate drugs discovered during preclinical screenings successfully advances to clinical trials (Biju, Priya, and Francis, 2023; Cha et al., 2025). Furthermore, only 3.4% of cancer drugs that passed Phase III clinical trials are commercially employed in clinical settings (Peng et al., 2025). Why?
The Convenience of the 2-Dimensional (2D) Monolayer Model – At A Cost
For decades, cancer researchers have been utilising the 2D monolayer cell culture model as an in vitro (in an artificial environment outside a living organism) approach in the laboratory to understand cancer better andidentify potential anticancer agents. This model is not only easy to establish and manipulate but also cost-effective, reproducible, and can be conveniently scaled and standardised for high-throughput screening (HTS) (Biju, Priya, and Francis, 2023; Cha et al., 2025; Zhou et al., 2025).
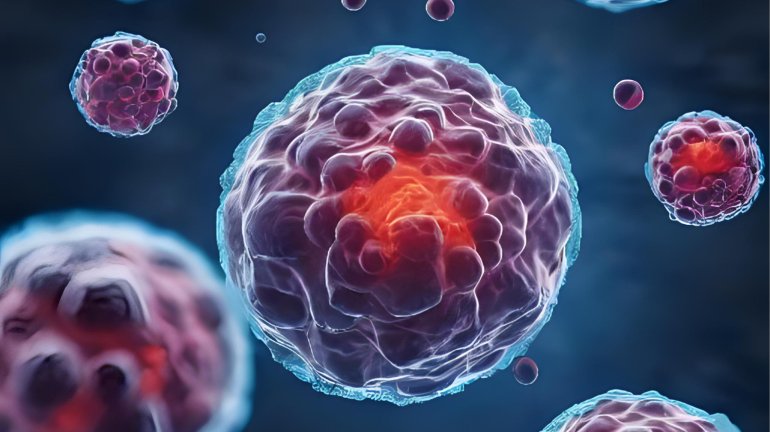
However, there is a significant fundamental flaw – unlike the monolayer culture, cancer cells, especially those in solid tumours, are often found in a three-dimensional (3D) arrangement, which determines how they behave and interact with their microenvironment (Biju, Priya, and Francis, 2023). Therefore, erratic cell behaviour and a lack of cell interaction between them and with the microenvironment in the monolayer model cause misleading responses to drugs, often false positives, due to differences in gene and protein expressions as well as the signalling pathways involved (Zhou et al., 2025; Biju, Priya, & Francis, 2023). To minimise or eliminate the inaccuracy of drug sensitivity, an increasing number of researchers have opted for the 3D cell culture technique as an in vitro model for drug testing in cancer research.
The Rise of the 3D Cell Culture Model – A Closer Look at Reality
To avoid inaccuracy in drug efficacy and toxicity, a 3D cell culture model is preferred due to its ability to recapitulate the ‘real’ physiological setting of tumours. This includes the presence of diffusion gradients of nutrients, oxygen, and waste, as well as interactions between cells and with the tumour microenvironment (TME), including the endothelial cells, extracellular matrix (ECM), immune and stromal cells (Cha et al., 2025; Marconi et al., 2025; Peng et al., 2025). These characteristics are crucial in determining tumour behaviour, particularly its aggressiveness and resistance to drugs (Zhou et al., 2025).
The most common type of 3D cell culture is the spheroid, which can be developed with or without scaffolds using various approaches (Cha et al., 2025). However, unlike scaffold-based methods that utilise collagen, matrigel, or synthetic polymers, scaffold-free approaches lack cell-ECM interactions. Regardless, the diffusion gradients are still present in both and are therefore superior to the monolayer model in predicting drug penetration. Furthermore, co-culturing cancer cells with non-cancerous components in the TME, such as endothelial and stromal cells, also allows for a better replication of the ‘real’ in vivo setting of tumours (Peng et al., 2025).
Despite their advantages, accuracy remains a primary challenge, as there are highly variable methods for developing 3D cell culture models, which may result in different cellular behaviours and interactions (Biju, Priya, and Francis, 2023). As such, microfluidic-based organ-on-a-chip models, patient-derived tumour organoids (PDTOs) and 3D bioprinting have been developed to recreate organ-like structures and have proven to provide more accurate responses to drugs (Cha et al., 2025; Zhou et al., 2025; Marconi et al., 2025; Peng et al., 2025). However, it is important to note that reproducibility remains a challenge due to the lack of standardised 3D models and the incompatibility of current evaluation methods (Biju, Priya, and Francis, 2023; Marconi et al., 2025). Nevertheless, 3D culture models are promising as ideal preclinical models for anticancer drug screening, thereby eliminating the ethical concerns associated with using animals for this purpose (Biju, Priya, and Francis, 2023). As the pharmaceutical industry seeks to improve translational success, embracing 3D culture technologies may be key to unlocking more effective and safer therapies, inspiring researchers to continue their efforts in cancer research.
References